MexBrain, a French startup based in Lyon, is launching a European clinical trial in the first semester of 2022 to help the most acute Wilson’s Disease patients.
The objective of this clinical trial will be to show a safe and strong extraction of copper and other toxic metals from the blood of Wilson’s Disease patients. It is not conceived as a replacement of existing treatments but as an addon to remove safely, quickly, and efficiently a large quantity of toxic metals from the blood.
Using a dialysis machine (widely available in hospitals and clinics), MexBrain’s product will act as a very strong chelator without the side effects of oral drugs. Its aim is to increase the chelation and extraction of copper from Wilson’s Disease patients. Here is a picture of how it would work:
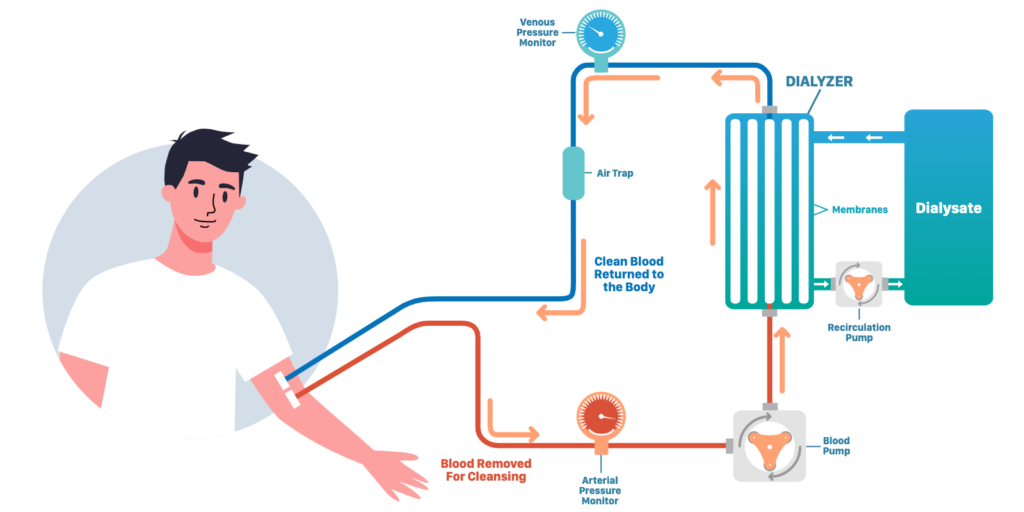
A central catheter would be inserted (same system as dialysis) in one of your veins in the leg or the chest. This small operation would take 10-15 minutes, and is performed by a nurse or doctor. Then a 4-hour session with a dialysis machine would be run, where you stay either in bed or seated and can watch TV, browse internet, read or just rest. MexBrain’s product is designed to stay in the dialysate (fluid on the right of the picture) and will never get in direct contact with your blood which is moving through the machine.
The clinical plan aims at up to 9 sessions per patient during one single week to extract sufficient copper from your body. It can be stopped at any time if you feel uncomfortable. You would have to stay close to the hospital for a period of 5 days, all expenses will be fully paid by MexBrain.
The clinical trial will start in 2022 in France, Germany, Austria, Spain, Poland and Denmark.
If you have a high free copper concentration in the blood, or display strong hepatic and/or neurologic symptoms, you might be eligible for this clinical trial. Please contact your doctor if you’re interested or directly MexBrain at patient@mexbrain.com